Today, when researchers spend long hours in the lab performing tricky experiments, they might listen to music or podcasts to get them through the day. But in the early years of neuroscience, hearing was an essential part of the process. To figure out what neurons cared about, researchers would translate the near-instantaneous signals they send, called “spikes,” into sound. The louder the sound, the more often the neuron was spiking—and the higher its firing rate.
“You can just hear how many pops are coming out of the speaker, and if it’s really loud or really quiet,” says Joshua Jacobs, associate professor of biomedical engineering at Columbia University. “And that’s a really intuitive way to see how active a cell is.”
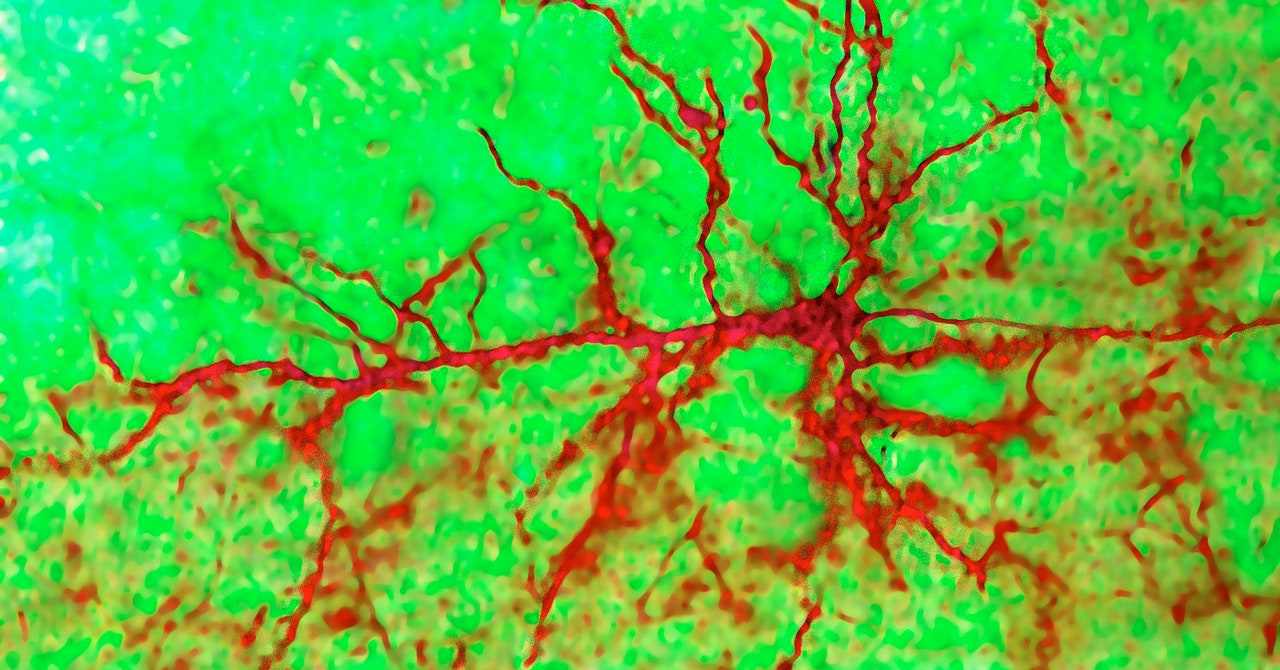
Neuroscientists don’t depend on sound anymore; they can record spikes with precision using implanted electrodes and computer software. To describe a neuron’s firing rate, a neuroscientist will choose a time window—say, 100 milliseconds—and see how many times it fires. Through firing rates, scientists have uncovered much of what we know about how the brain works. Examining them in a deep region of the brain called the hippocampus, for example, led to the discovery of place cells—cells that become active when an animal is in a particular location. This 1971 discovery won neuroscientist John O’Keefe a 2014 Nobel Prize.
Firing rates are a useful simplification; they show a cell’s overall activity level, although they sacrifice precise information about spike timing. But individual sequences of spikes are so intricate, and so variable, that it can be hard to figure out what they mean. So focusing on firing rates often comes down to pragmatics, says Peter Latham, a professor in the Gatsby Computational Neuroscience Unit at University College London. “We never have enough data,” Latham says. “Every single trial is completely different.”
But that doesn’t mean studying spike timing is pointless. Though interpreting a neuron’s spikes is tricky, finding meaning in those patterns is possible, if you know what you’re looking for.
That’s what O’Keefe was able to do in 1993, more than two decades after he discovered place cells. By comparing the timing of when these cells fired to local oscillations—overall wavelike patterns of activity in a brain region—he discovered a phenomenon called “phase precession.” When a rat is at a particular location, that neuron will fire around the same time that other nearby neurons are most active. But as the rat keeps moving, that neuron will fire a little bit before, or a little bit after, the peak activity of its neighbors. When a neuron becomes increasingly out of sync with its neighbors over time, it’s exhibiting phase precession. Eventually, since the background brain activity follows a repetitive, up-and-down pattern, it will get back in sync with it, before starting the cycle again.
Since O’Keefe’s discovery, phase precession has been intensively studied in rats. But no one knew for sure if it happens in humans until May, when Jacobs’ team published in the journal Cell the first evidence of it in the human hippocampus. “This is good news, because things are falling in place across different species, different experimental conditions,” says Mayank Mehta, a prominent phase precession researcher at UCLA, who was not involved in the study.
The Columbia University team made their discovery via decade-old recordings from the brains of epileptic patients that tracked neural activity as the patients navigated a virtual environment on a computer. Epilepsy patients are often recruited for neuroscience research because their treatment can involve surgically implanted deep brain electrodes, which give scientists a unique opportunity to eavesdrop on the firing of individual neurons in real time.